Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
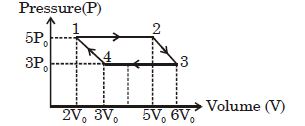
- 1A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are :View Solution
${P_A} = 3 \times {10^4}Pa,\;{P_B} = 8 \times {10^4}Pa$ and ${V_A} = 2 \times {10^{ - 3}}{m^3},\;{V_D} = 5 \times {10^{ - 3}}{m^3}$
In process $AB$, $600 J$ of heat is added to the system and in process $BC, 200 J $ of heat is added to the system. The change in internal energy of the system in process $ AC$ would be ...... $J$
- 2$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 3An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
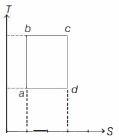
- 4An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram isView Solution
- 5The pressure in the tyre of a car is four times the atmospheric pressure at $300 K$. If this tyre suddenly bursts, its new temperature will be $(\gamma = 1.4)$View Solution
- 6The pressure of an ideal gas varies with volume as $P = \alpha V,$ where $\alpha $ is a constant. One mole of the gas is allowed to undergo expansion such that its volume becomes $'m'$ times its initial volume. The work done by the gas in the process isView Solution
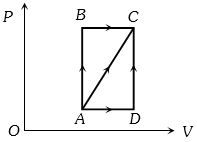
- 7One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
- 8Efficiency of Carnot engine is $100\%$ ifView Solution
- 9View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 10View SolutionEntropy of a system decreases .........