The efficiency of carnot engine is $50\%$ and temperature of sink is $500\,K$ . If temperature of source is kept constant and its efficiency raised to $60\%$ , then the required temperature of the sink will be .... $K$
Medium
$\mathrm{T}_{\sin \mathrm{k}}=500$
$\eta=50 \%$
$\mathrm{T}_{\text {source }}=1000 \mathrm{k}$
Now $\eta_{2}=60 \%$
$\frac{60}{100}=\frac{1000-\mathrm{T}_{2}}{1000}$
$\mathrm{T}_{2}=400 \mathrm{k}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
- 2Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ isView Solution
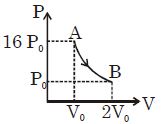
- 3Find $V_B = ?$View Solution
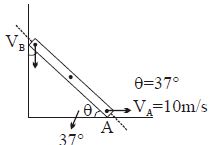
- 4An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution
- 5View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
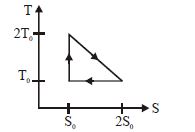
- 6View SolutionIn the following figures heat is absorbed by the gas
- 7The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
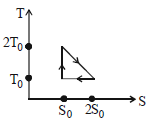
- 8View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
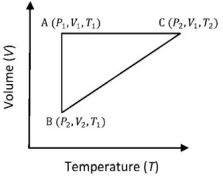
- 9A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
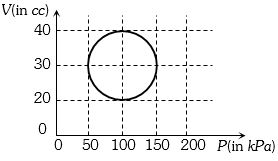
- 10View SolutionA system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is