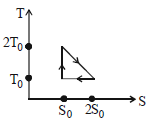
${\mathrm{Q}_{2}=\mathrm{T}_{0}\left(2 \mathrm{S}_{0}-\mathrm{S}_{0}\right)=\mathrm{T}_{0} \mathrm{S}_{0} \text { and } \mathrm{Q}_{3}=0}$
${\eta=\frac{\mathrm{W}}{\mathrm{Q}_{1}}=\frac{\mathrm{Q}_{1}-\mathrm{Q}_{2}}{\mathrm{Q}_{1}}} $
${=1-\frac{\mathrm{Q}_{2}}{\mathrm{Q}_{1}}=1-\frac{\mathrm{T}_{0} \mathrm{S}_{0}}{\frac{3}{2} \mathrm{T}_{0} \mathrm{S}_{0}}=\frac{1}{3}}$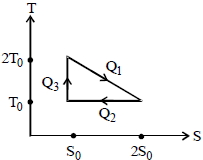
Download our appand get started for free
Similar Questions
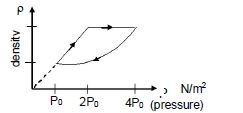
- 1An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 2Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
- 3The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
- 4An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
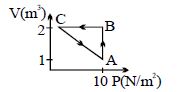
- 5View SolutionThe state of a thermodynamic system is represented by
- 6One mole of an ideal gas is contained with in a cylinder by a frictionless piston and is initially at temperature $T$. The pressure of the gas is kept constant while it is heated and its volume doubles. If $R$ is molar gas constant, the work done by the gas in increasing its volume isView Solution
- 7Starting at temperature $300\; \mathrm{K},$ one mole of an ideal diatomic gas $(\gamma=1.4)$ is first compressed adiabatically from volume $\mathrm{V}_{1}$ to $\mathrm{V}_{2}=\frac{\mathrm{V}_{1}}{16} .$ It is then allowed to expand isobarically to volume $2 \mathrm{V}_{2} \cdot$ If all the processes are the quasi-static then the final temperature of the gas (in $\left. \mathrm{K}\right)$ is (to the nearest integer)View Solution
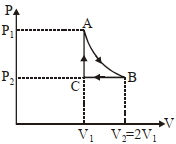
- 8$n$ mole a perfect gas undergoes a cyclic process $ABCA$ (see figure) consisting of the following processes.View Solution
$A \rightarrow B :$ Isothermal expansion at temperature $T$ so that the volume is doubled from $V _{1}$ to $V _{2}=2 V _{1}$ and pressure changes from $P _{1}$ to $P _{2}$
$B \rightarrow C :$ Isobaric compression at pressure $P _{2}$ to initial volume $V _{1}$
$C \rightarrow A$ : Isochoric change leading to change of pressure from $P _{2}$ to $P _{1}$
Total workdone in the complete cycle $ABCA$ is
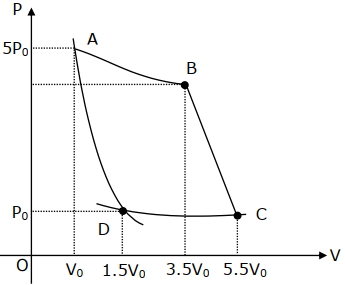
- 9In the reported figure, there is a cyclic process $ABCDA$ on a sample of $1\, {mol}$ of a diatomic gas. The temperature of the gas during the process ${A} \rightarrow {B}$ and ${C} \rightarrow {D}$ are ${T}_{1}$ and ${T}_{2}\left({T}_{1}\,>\,{T}_{2}\right)$ respectively.View Solution
Choose the correct option out of the following for work done if processes $B C$ and $D A$ are adiabatic.
- 10In a heat engine, the temperature of the source and sink are $500\, K$ and $375\, K$. If the engine consumes $25\times10^5\, J$ per cycle, the work done per cycle isView Solution
