A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is
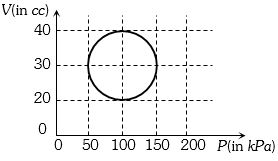

Medium
(b) In cyclic process $\Delta Q =$ Work done = Area inside the closed curve.
Treat the circle as an ellipse of area $ = \frac{\pi }{4}({P_2} - {P_1})\,({V_2} - {V_1})$
$\Rightarrow \Delta Q = \frac{\pi }{4}\{ (150 - 50) \times {10^3}\} = \frac{\pi }{2}J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
- 2A thermally insulated container is divided into two parts by a screen. In one part the pressure and temperature are $P$ and $T$ for an ideal gas filled. In the second part it is vacuum. If now a small hole is created in the screen, then the temperature of the gas willView Solution
- 3View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 4A gas is suddenly compressed to $1/4$ th of its original volume at normal temperature. The increase in its temperature is ....... $K$ $(\gamma = 1.5)$View Solution
- 5If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$View Solution
- 6A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
- 7Initial pressure and volume of a gas are $ P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $ V$. The final pressure of gas will beView Solution
- 8An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution
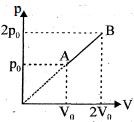
- 9A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 10A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process? $( J =4.18$ $Joules / cal )$ (in joule)View Solution
