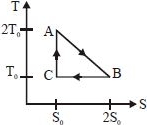
The temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be comeView Solution
- 2Slope of isotherm for a gas (having $\gamma=\frac{5}{3}$ ) is $3 \times 10^5 \,N / m ^2$. If the same gas is undergoing adiabatic change then adiabatic elasticity at that instant is ........... $\times 10^5 N / m ^2$View Solution
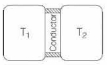
- 3A thin piece of thermal conductor of constant thermal conductivity insulated on the lateral sides connects two reservoirs which are maintained at temperatures $T_{1}$ and $T_{2}$ as shown in the figure alongside. Assuming that the system is in steady state, which of the following plots best represents the dependence of the rate of change of entropy on the ratio of $T_{1} / T_{2}$ ?View Solution
- 4One mole of an ideal gas at temperature $T_1$ expends according to the law $\frac{P}{{{V^2}}} =a$ (constant). The work done by the gas till temperature of gas becomes $T_2 $ isView Solution
- 5View SolutionIrreversible process is
- 6A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
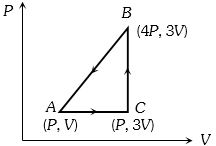
- 7A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
- 8A total of $48 \mathrm{~J}$ heat is given to one mole of helium kept in a cylinder. The temperature of helium increases by $2^{\circ} \mathrm{C}$. The work done by the gas is : (Given, $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$.)View Solution
- 9Starting with the same initial conditions, an ideal gas expands from volume $V_{1}$ to $V_{2}$ in three different ways. The work done by the gas is $W_{1}$ if the process is purely isothermal. $W _{2}$. if the process is purely adiabatic and $W _{3}$ if the process is purely isobaric. Then, choose the coned optionView Solution
- 10A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process? $( J =4.18$ $Joules / cal )$ (in joule)View Solution