The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$
Easy
(c) $\Delta Q = \Delta U + \Delta W$$ \Rightarrow \Delta U = \Delta Q - \Delta W = 2240 - 168 = 2072\;J$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
- 2$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 3Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
- 4During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
- 5The temperature of a hypothetical gas increases to $\sqrt 2 $ times when compressed adiabatically to half the volume. Its equation can be written asView Solution
- 6View SolutionCan two isothermal curves cut each other
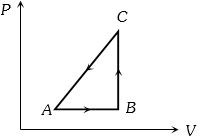
- 7The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
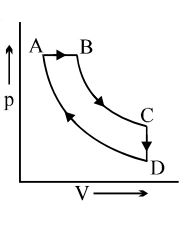
- 8A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
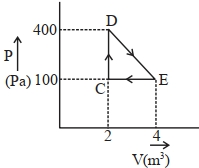
- 9A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
- 10Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\epsilon_1$ and $\epsilon_2$, respectively. The net efficiency of the combination is given by :View Solution