A certain amount of gas of volume $V$ at $27^{o}\,C$ temperature and pressure $2 \times 10^{7} \;Nm ^{-2}$ expands isothermally until its volume gets doubled. Later it expands adiabatically until its volume gets redoubled. The final pressure of the gas will be (Use $\gamma=1.5$ )
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
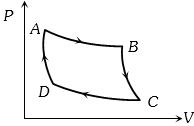
- 1An ideal gas of mass $m$ in a state $A$ goes to another state $B$ via three different processes as shown in figure. If $Q_{1}, Q_{2}$ and $Q_{3}$ denote the heat absorbed by the gas along the three paths, thenView Solution
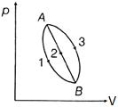
- 2A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
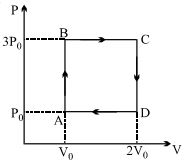
- 3An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
- 4View SolutionWork done in the cyclic process shown in figure is ...........
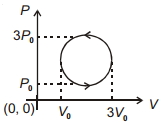
- 5The coefficient of performance of a refrigerator is $5$ . If the temperature inside freezer is $-20\,^oC$ , the temperature of the surroundings to which it rejects heat is ....... $^oC$View Solution
- 6View SolutionThe state of a thermodynamic system is represented by
- 7The $P-V$ diagram of $2\,g$ of helium gas for a certain process $A$ $\to$ $B$ is shown in the figure. What is the heat given to the gas during the process $A$ $\to$ $B$?View Solution
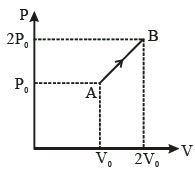
- 8Work done for the process shown in the figure is ............ $J$View Solution
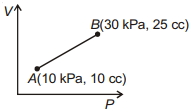
- 9An air bubble of volume $v _0$ is released by a fish at a depth $h$ in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density) of water is $\rho$View Solution
- 10Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct