The specific heat relation for ideal gas is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 2Molar specific heat at constant volume is ${C_v}$ for a monoatomic gas isView Solution
- 3View SolutionMaxwell distribution function is shown in figure for different gases, which of the following is correct matching?
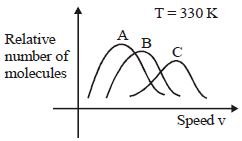
- 4View SolutionSelect the incorrect relation. (Where symbols have their usual meanings)
- 5The molar heat capacity in a process of a diatomic gas if it does a work of $\frac{Q}{4}$ when a heat of $Q$ is supplied to it isView Solution
- 6The average distance between molecules of an ideal gas at $STP$ is approximately of the order ofView Solution
- 7View SolutionThe number of translational degrees of freedom for a diatomic gas is
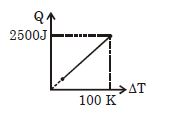
- 8One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
- 9View SolutionA closed compartment containing gas is moving with some acceleration in horizontal direction. Neglect effect of gravity. Then the pressure in the compartment is
- 10Consider a mixture of $n$ moles of helium gas and $2 n$ moles of oxygen gas (molecules taken to be rigid) as an ideal gas. Its $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}$ value will beView Solution
