The heat capacity of one mole an ideal is found to be $C_V=3 R(1+a R T) / 2$, where $a$ is a constant. The equation obeyed by this gas during a reversible adiabatic expansion is
KVPY 2016, Advanced
(A)
From the given data,
$C _{ V }=\frac{3 R ( a + aRT )}{2}$
So,
$C _{ p }= C _{ V }+ R =\frac{3 R ( a + aRT )}{2}+ R =\frac{3 R ( a + aRT )+2 R }{2}$
So,
$\gamma=\frac{C_{ P }}{C_{ V }}-1=\frac{\frac{3 R(a+a R T)+2 R}{2}}{\frac{3 R(a+a R T)}{2}}-1=\frac{2}{3(1+a R T)}=\frac{2}{3}(1+a R T)^{-1}$
Now, by putting this value of $\gamma-1$ in the adiabatic expression formula, we get $TV ^{\gamma-1}=$ constant $\Rightarrow TV ^{3 / 2} e ^{ aRT }=$ constant (we get this by binomial expansion).
Hence proved.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 2View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
- 3View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
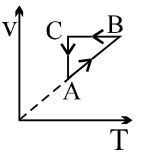
- 4The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
- 5Three Carnot engines operate in series between a heat source at a temperature $T_1$ and a heat sink at temperature $T_4$ (see figure). There are two other reservoirs at temperature $T_2$ and $T_3$, as shown, with $T_1 > T_2 > T_3 > T_4$. The three engines are equally efficient ifView Solution
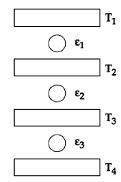
- 6View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
- 7View SolutionCofficient of performance of refigerator is
- 8If during an adiabatic process the pressure of mixture of gases is found to be proportional to square of its absolute temperature. The ratio of $C_p / C_v$ for mixture of gases is .........View Solution
- 9A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 10View SolutionWhich is the correct statement
