If during an adiabatic process the pressure of mixture of gases is found to be proportional to square of its absolute temperature. The ratio of $C_p / C_v$ for mixture of gases is .........
Medium
(a)
$P \propto T^2$
$P T^{-2}=$ constant $\quad$ compare with $P T^{\left(\frac{\gamma}{1-\gamma}\right)}=$ constant
$\frac{C_P}{C_V}=\gamma=2$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 2An ideal gas follows a process described by the equation $PV ^2= C$ from the initial $\left( P _1, V _1, T _1\right)$ to final $\left(P_2, V_2, T_2\right)$ thermodynamics states, where $C$ is a constant. ThenView Solution
- 3If during an adiabatic process the pressure of mixture of gases is found to be proportional to square of its absolute temperature. The ratio of $C_p / C_v$ for mixture of gases is .........View Solution
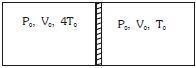
- 4Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 5View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 6$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
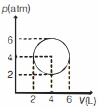
- 7An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
- 8$\Delta U + \Delta W = 0$ is valid forView Solution
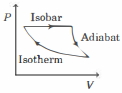
- 9The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
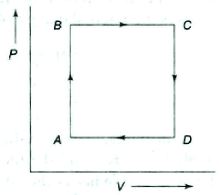
- 10A thermodynamic process is the pressure and volumes corresponding to some points in the figure are, $P_A = 3 \times 10^4 Pa$, $V_A = 2 \times 10^{-3}\, m^3$, $P_B = 8 \times 10^4 Pa$, $V_D = 5 \times 10^{-3}\,m^3$. In process $AB, 600\, J$ of heat and in process $BC, 200\, J$ of heat is added to the system. The change in the internal energy in process $AC$ would be .... $J$View Solution