A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases is
Medium
(b) $(i)$ Case $ \to $Volume = constant $ \Rightarrow \int_{}^{} {PdV} = 0$
$(ii)$ Case$ \to P =$ constant $ \Rightarrow \int_{\,{V_1}}^{\,2{V_1}} {PdV} = P\int_{\,{V_1}}^{\,2{V_1}} {dV = P{V_1}} $
$(ii)$ Case$ \to P =$ constant $ \Rightarrow \int_{\,{V_1}}^{\,2{V_1}} {PdV} = P\int_{\,{V_1}}^{\,2{V_1}} {dV = P{V_1}} $
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two Carnot engines $A$ and $B$ are operated in series. The first one, $A,$ receives heat at $T_1(= 600\,K)$ and rejects to a reservoir at temperature $T_2.$ The second engine $B$ receives heat rejected by the first engine and, in turns, rejects to a heat reservoir at $T_3 (=400\,K).$ Calculate the temperature $T_2$ if the work outputs of the two engines are equal ..... $K$View Solution
- 2$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
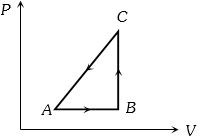
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 3The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
- 4A diatomic gas initially at $18^o C$ is compressed adiabatically to one-eighth of its original volume. The temperature after compression will beView Solution
- 5$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 6An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
- 7A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
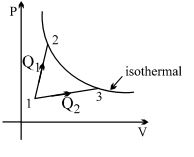
- 8A gas takes part in two processes in which it is heated from the same initial state $1$ to the same final temperature. The processes are shown on the $P-V$ diagram by the straight line $1-2$ and $1-3$. $2$ and $3$ are the points on the same isothermal curve. $Q_1$ and $Q_2$ are the heat transfer along the two processes. ThenView Solution
- 9The efficiency of an ideal heat engine working between the freezing point and boiling point of water, is ........ $\%$View Solution
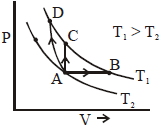
- 10Three different processes that can occur in an ideal monoatomic gas are shown in the $P$ vs $V$ diagram. The paths are labelled as $A \rightarrow B, A \rightarrow C$ and $A \rightarrow D .$ The change in internal energies during these process are taken as $E _{ AB }, E _{ AC }$ and $E _{ AD }$ and the work done as $W _{ AB }$ $W _{ AC }$ and $W _{ AD }$View Solution
The correct relation between these parameters are