A process can be reversible if
Easy
(c)
A quasi-static process is a thermodynamic process that happens slowly enough for the system to remain in internal equilibrium. it means the change in one step system can be reversed after taking that step at any point of the process. The process should also be non-dissipative, i.e. there should not be any friction or air drag which results in loss of energy of system.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an abosolute temperature $T$ and rejects heat to a sink at a temperature of $T/3.$ The amount of heat rejected isView Solution
- 2If $Q , E$ and $W$ denote respectively the heat added, change in internal energy and the work done by a closed cycle process, thenView Solution
- 3One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
- 4View SolutionIn a cyclic process, work done by the system is
- 5$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
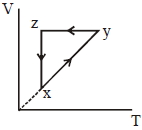
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 6A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 7A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
- 8Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 9A gas can be taken from $A$ to $B$ via two different processes $ACB$ and $ADB$. When path $ACB$ is used $60\, J$ of heat flows into the system and $30\, J$ of work is done by the system. If path $ADB$ is used work down by the system is $10\, J$. the heat flow into the system in path $ADB$ is ..... $J$View Solution
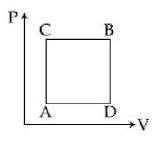
- 10An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
