Reagents used for industrial preparation of methanol are$:$
Carbon monoxide and hydrogen react over a catalyst to produce methanol.
Today, the most widely used catalyst is a mixture of copper and zinc oxides,supported on alumina.
At $5–10 \ \ce{MPa (50–100 atm)}$ and $250^\circ C (482^\circ F),$ the reaction is characterized by high selectivity $(>99.8\%).$
$\ce{CO + 2H_2 \rightarrow CH_{3}OH}$
Today, the most widely used catalyst is a mixture of copper and zinc oxides,supported on alumina.
At $5–10 \ \ce{MPa (50–100 atm)}$ and $250^\circ C (482^\circ F),$ the reaction is characterized by high selectivity $(>99.8\%).$
$\ce{CO + 2H_2 \rightarrow CH_{3}OH}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionStrength of acidity is in order:
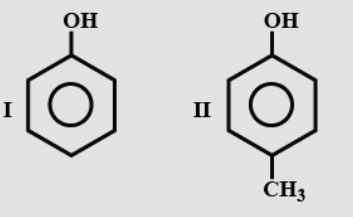
- 2View SolutionWhich is the only alcohol that can be prepared by the indirect hydration of alken?
- 3View SolutionWhich of the following alcohol does not give a stable compound on dehydration?
- 4View SolutionMark the correct order of decreasing acid strength of the following compounds.
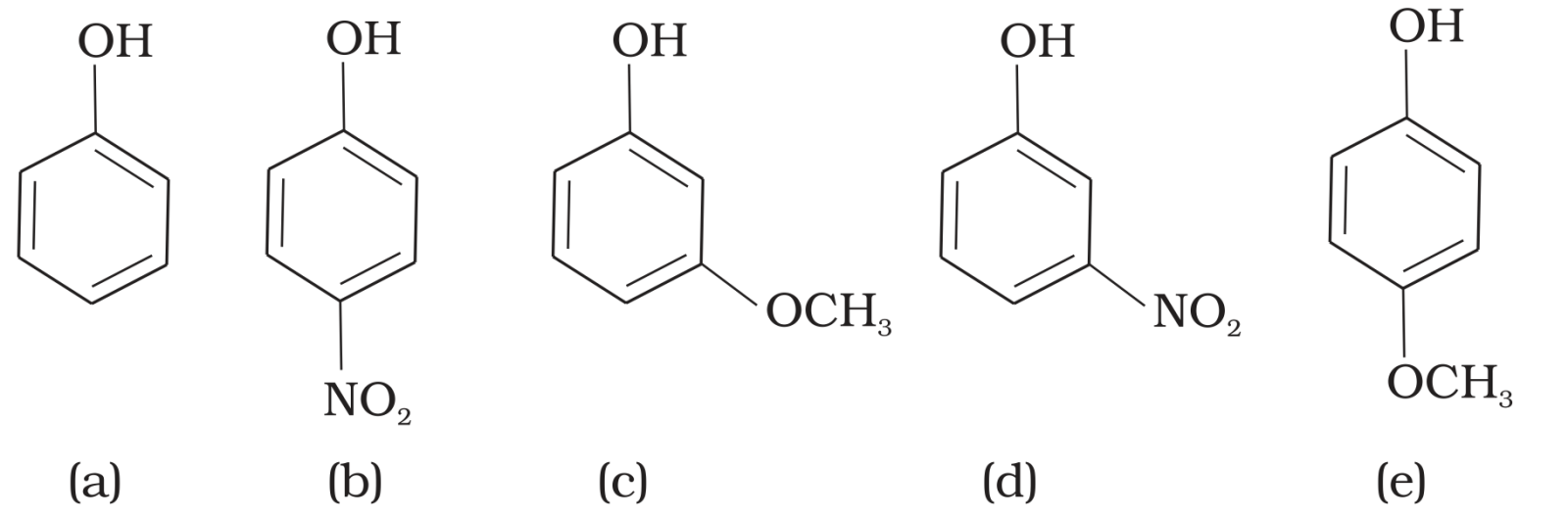
- 5$\ce{IUPAC}$ name of $m-$cresol is $.......$View Solution
- 6The compound which gets dissolved in water is$:$View Solution
- 7View SolutionWhich of the following substance will increase the acidity of phenol?
- 8View SolutionWhich of the following is a primary alcohol?
- 9Phenol can be distinguished from ethanol by the reactions with $.........$View Solution
- 10When $3-$methylbutan$-2-$ol is treated with $\ce{HBr},$ the following reaction takes place. What are the correct steps for formation of the above product?View Solution
