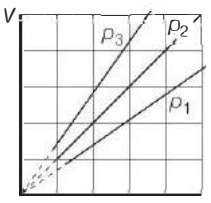
The state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?

KVPY 2015, Diffcult
(b)
Equation of state for ideal gas is $p V=n R T \Rightarrow V=\frac{n R}{p} \cdot T$
This is equation of line with slope $=\frac{n R}{p}$
$\Rightarrow \quad$ Slope $\propto \frac{1}{p}$
So, from graph, $p_3 < p_2 < p_1$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
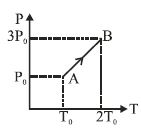
- 1Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
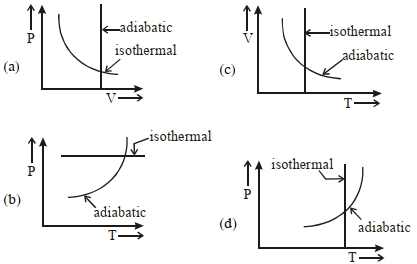
- 2View SolutionWhich one is the correct option for the two different thermodynamic processes ?
- 3Two moles of an ideal monoatomic gas at ${27^o}C$ occupies a volume of $V.$ If the gas is expanded adiabatically to the volume $2V,$ then the work done by the gas will be ....... $J$ $[\gamma = 5/3,\,R = 8.31J/mol\,K]$View Solution
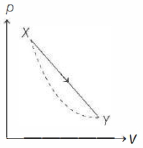
- 4In the $p-V$ diagram below, the dashed curved line is an adiabat.For a process that is described by a straight line joining two points $X$ and $Y$ on the adiabat (solid line in the diagram) heat is (Hint consider the variation in temperature from $X$ to $Y$ along the straight line)View Solution
- 5$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 6$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
- 7In the $P-V$ diagram shown, the gas does $5\, J$ of work in isothermal process $ab$ and $4\,J$ in adiabatic process $bc$. .... $J$ will be the change in internal energy of the gas in straight path $c$ to $a$ ?View Solution
- 8View SolutionIn which thermodynamic process, volume remains same
- 9An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
- 10In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
