The latent heat of vaporization of water is $2240 \,J/gm$. If the work done in the process of vaporization of $1\, gm$ is $168\, J$, then increase in internal energy is .... $J$
Medium
$\mathrm{L}=2240 \mathrm{J}, \mathrm{m}=1 \mathrm{gm}$
$\mathrm{d} \mathrm{W}=168 \mathrm{J}$
$\mathrm{dQ}=\mathrm{mL}=\mathrm{dU}+\mathrm{d} \mathrm{W}$
or $1 \times 2240=d U+168$
$\mathrm{dU}=2072 \mathrm{J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
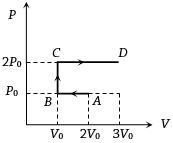
- 1$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ isView Solution
- 2The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 3Work done by air when it expands from $50\, litres$ to $150\, litres$ at a constant pressure of $2$ atmosphere isView Solution
- 4Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
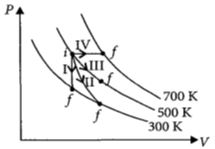
- 5Thermodynamic processes are indicated in the following diagram.View Solution
Match the following
$\begin{array}{|l|l|} \hline Column\,\,-\,\,1 & Column\,\,-\,\,2 \\ \hline P\,:\,Process\,\,-\,\,I & \,\,A\,\,:\,\,Adiabatic \\ \hline Q\,:\,Process\,\,-\,\,II & \,\,B\,\,:\,\,Isobaric \\ \hline R\,:\,Process\,\,-\,\,III & \,\,C\,\,:\,\,Isochoric \\ \hline S\,:\,Process\,\,-\,\,IV & \,\,D\,\,:\,\,Isothermal \\ \hline \end{array}$
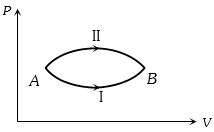
- 6A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
- 7A van der Waal's gas obeys the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$. Its internal energy is given by $U=C T-\frac{n^2 a}{V}$. The equation of a quasistatic adiabat for this gas is given byView Solution
- 8View SolutionIf a system undergoes contraction of volume then the work done by the system will be
- 9Find the amount of work done to increase the temperature of one mole of an ideal gas by $30^o\ C$ if it is expanding under the condition .... $J$View Solution
$V\propto {T^{\frac{2}{3}}}$ $[R = 1.99\ cal/mol-K]$
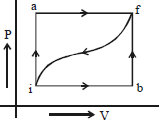
- 10When a system is taken from a state $i$ to $f$ along the path $iaf$ (as shown in the figure). $Q = 50\, cal$ and $W = 20\, cal$ ; along path $ibf,$ $Q = 36\, cal.$View Solution
$(i)$ What is $W$ along path $ibf$ ?
$(ii)$ If $W = 13$ cal for path $fi$, what is $Q$ for the path $fi$ ?
$(iii)$ Take $E_{int,i} = 10\,\, cal$ then what is $E_{int,f}$ ?