$(i)$ What is $W$ along path $ibf$ ?
$(ii)$ If $W = 13$ cal for path $fi$, what is $Q$ for the path $fi$ ?
$(iii)$ Take $E_{int,i} = 10\,\, cal$ then what is $E_{int,f}$ ?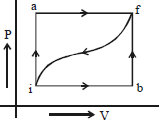
$Q=50 \mathrm{cal}$
$\mathrm{W}=20 \mathrm{cal}$
According ot I law of thermodynamics,
$\mathrm{dQ}=\mathrm{dU}+\mathrm{d} \mathrm{W}$
$\text { or } \mathrm{dU}=\mathrm{dQ}-\mathrm{dW}=50-20=30 \mathrm{cal}$
(1) For path iaf
$Q=36\,cal$
${W=?}$
$\mathrm{dU}=30 \mathrm{cal}($ since internal energy depends only on the initial and final positions of the system).
$\therefore W=Q-d U=36-30=6 \mathrm{cal}$
$\begin{aligned} \text { (u) } \mathrm{W}=-13 \mathrm{cal} \\ \mathrm{d} \mathrm{U}=-30 \mathrm{cal} \\ \mathrm{Q}=? \end{aligned}$
$\therefore \mathrm{Q}=\mathrm{dU}+\mathrm{W}=-43 \mathrm{cal}$
(iii) $\quad \mathrm{E}_{\text {int }, \mathrm{f}}=\mathrm{E}_{\text {int }, i}+\Delta \mathrm{U}=10 \mathrm{cal}+30 \mathrm{cal} .=40$
cal.
Download our appand get started for free
Similar Questions
- 1The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
- 2A diatomic gas $(\gamma=1.4)$ does $200 \mathrm{~J}$ of work when it is expanded isobarically. The heat given to the gas in the process is:View Solution
- 3$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 4An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
- 5A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution
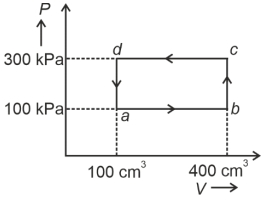
- 6If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
- 7On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
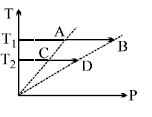
- 8For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
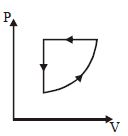
- 9View SolutionWhich one is the correct option for the two different thermodynamic processes ?
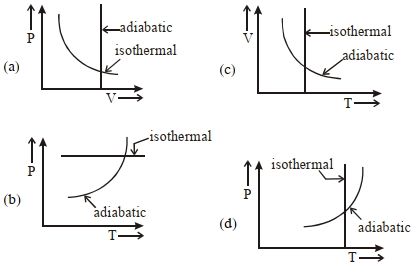
- 10$PV$ curve for the process whose $VT$ curve isView Solution