Thermodynamic processes are indicated in the following diagram.
Match the following
$\begin{array}{|l|l|} \hline Column\,\,-\,\,1 & Column\,\,-\,\,2 \\ \hline P\,:\,Process\,\,-\,\,I & \,\,A\,\,:\,\,Adiabatic \\ \hline Q\,:\,Process\,\,-\,\,II & \,\,B\,\,:\,\,Isobaric \\ \hline R\,:\,Process\,\,-\,\,III & \,\,C\,\,:\,\,Isochoric \\ \hline S\,:\,Process\,\,-\,\,IV & \,\,D\,\,:\,\,Isothermal \\ \hline \end{array}$
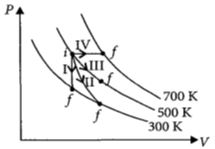
NEET 2017, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. Adiabatic constant for the gas is $\gamma=3 / 2$. The work done by the gas in the process is : $(\mu=1 \mathrm{~mole})$View Solution
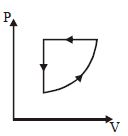
- 2For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
- 3A thermally insulated rigid container contains an ideal gas heated by a filament of resistance $100 \,\Omega$ through a current of $1\,A$ for $5$ min then change in internal energy is...... $kJ$View Solution
- 4View SolutionIn a cyclic process, the internal energy of the gas
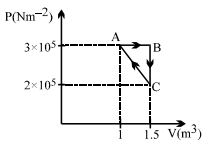
- 5Considere the thermodynamics cycle shown on $PV$ diagram. The process $A \rightarrow B$ is isobaric, $B \rightarrow C$ is isochoric and $C \rightarrow A$ is a straight line process. The following internal energy and heat are given $: \Delta U_{A \rightarrow B} = + 400\,\, kJ$ and $Q_{B \rightarrow C} = - 500\,\, kJ$ The heat flow in the process $Q_{C \rightarrow A}$ is ...... $kJ$View Solution
- 6View SolutionWhen an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is
- 7The efficiency of carnot engine is $50\%$ and temperature of sink is $500\;K$. If temperature of source is kept constant and its efficiency raised to $60\%$, then the required temperature of the sink will beView Solution
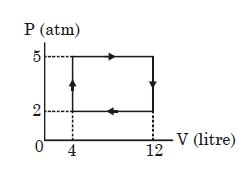
- 8View SolutionAn ideal gas undergoes a cyclic process as shown in diagram. The net work done by the gas in the cycle is
- 9A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
- 10View SolutionFor an ideal gas, in an isothermal process
