Which one is the correct option for the two different thermodynamic processes ?
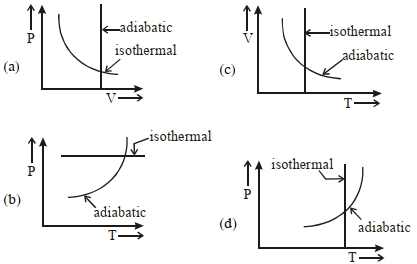

JEE MAIN 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
- 2A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$View Solution
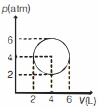
- 3An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
- 4View SolutionWhich is the correct statement
- 5An engine has an efficiency of $0.25$ when temperature of sink is reduced by $58\,^oC$, if its efficiency is doubled, then the temperature of the source is ..... $^oC$View Solution
- 6View SolutionWhich of the following is not thermodynamical function
- 7An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
- 8A balloon filled with helium $\left(32^{\circ} C \right.$ and $1.7\; atm$.) bursts. Immediately afterwards the expansion of helium can be considered asView Solution
- 9$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
- 10A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
