The pressure of an ideal gas varies with volume as $P = \alpha V,$ where $\alpha $ is a constant. One mole of the gas is allowed to undergo expansion such that its volume becomes $'m'$ times its initial volume. The work done by the gas in the process is
AIEEE 2012, Medium
$\text { Given } P=\alpha V $
$\text { Work done, } w=\int_{V}^{mV} P d V $
$=\int_{m}^{m V} a V d V=\frac{\alpha V^{2}}{2}\left(m^{2}-1\right)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe first operation involved in a Carnot cycle is
- 2If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, isView Solution
- 3An ideal refrigerator has a freezer at a temperature of $ - 13^\circ C.$ The coefficient of performance of the engine is $5.$ The temperature of the air (to which heat is rejected) will beView Solution
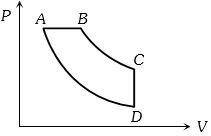
- 4View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 5Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
- 6A gas undergoes a change of state during which $100 J$ of heat is supplied to it and it does $20 J$ of work. The system is brought back to its original state through a process during which $20 J$ of heat is released by the gas. The work done by the gas in the second process is ....... $J$View Solution
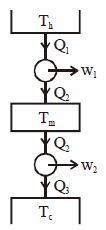
- 7Suppose that two heat engines are connected in series, such that the heat exhaust of the first engine is used as the heat input of the second engine as shown in figure. The efficiencies of the engines are $\eta_1$ and $\eta_2$, respectively. The net efficiency of the combination is given byView Solution
- 8The volume of a gas is reduced adiabatically to $\frac{1}{4}$ of its volume at $27°C$, if the value of $\gamma = 1.4,$ then the new temperature will beView Solution
- 9The amount of heat needed to raise the temperature of $4\, moles$ of a rigid diatomic gas from $0^{\circ} {C}$ to $50^{\circ} {C}$ when no work is done is ......${R}$ ($R$ is the universal gas constant)View Solution
- 10An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
