If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, is
AIPMT 1998, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 2View SolutionA gas is being compressed adiabatically. The specific heat of the gas during compression is
- 3View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 4A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
- 5The volume of a gas is reduced adiabatically to $\frac{1}{4}$ of its volume at $27°C$, if the value of $\gamma = 1.4,$ then the new temperature will beView Solution
- 6If $R =$ universal gas constant, the amount of heat needed to raise the temperature of $2$ mole of an ideal monoatomic gas from $273K$ to $373K$ when no work is done ...... $R$View Solution
- 7In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 8A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an abosolute temperature $T$ and rejects heat to a sink at a temperature of $T/3.$ The amount of heat rejected isView Solution
- 9View SolutionWhich one is the correct option for the two different thermodynamic processes ?
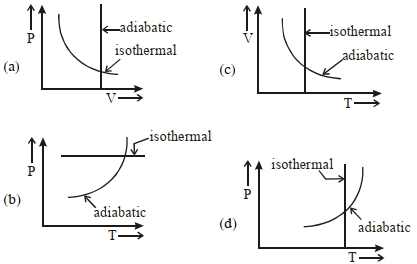
- 10In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
