The mean free path of molecules of a certain gas at $STP$ is $1500\,d$, where $d$ is the diameter of the gas molecules. While maintaining the standard pressure, the mean free path of the molecules at $373\,K$ is approximately $..........\,d$
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The following sets of values for ${C_V}$ and ${C_P}$ of a gas has been reported by different students. The units are $cal/gm-mole-K.$ Which of these sets is most reliableView Solution
- 2View SolutionMaxwell distribution function is shown in figure for different gases, which of the following is correct matching?
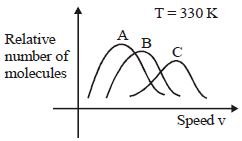
- 3View SolutionA gas is enclosed in a closed pot. On keeping this pot in a train moving with high speed, the temperature of the gas
- 4One mole of a monoatomic gas is mixed with three moles of a diatomic gas. The molecular specific heat of mixture at constant volume is $\frac{\alpha^{2}}{4} R J / mol\,K$; then the value of $\alpha$ will be $.......$ (Assume that the given diatomic gas has no vibrational mode.)View Solution
- 5If the r.m.s. speed of chlorine molecule is $490\,m / s$ at $27^{\circ}\,C$, the r.m.s. speed of argon molecules at the same temperature will be $......\,m/s$ (Atomic mass of argon $=39.9\,u$, molecular mass of chlorine $=70.9\,u )$View Solution
- 6The relation $C_p-C_V=R\left(C_p\right.$ and $C_V$ are the molar specific heats at constant pressure and volume) is exactly true forView Solution
- 7$70$ calories of heat are required to raise the temperature of $2$ moles of an ideal gas at constant pressure from $30°C$ to $35°C.$ The amount of heat required to raise the temperature of same gas through the same range $(30°C$ to $35°C)$ at constant volume ..... $cal$ $(R = 2 \,cal/mol/K)$View Solution
- 8The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
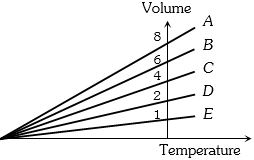
- 9The ratio of mean kinetic energy of hydrogen and nitrogen at temperature $300 \,K$ and $450\, K$ respectively isView Solution
- 10The relation between the gas pressure $P$ and average kinetic energy per unit volume $E$ isView Solution
