The specific heat of a gas
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The mean free path of molecules ofa gas, (radius $r$) is inversely proportional toView Solution
- 2For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
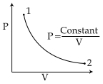
- 3Under an adiabatic process, the volume of an ideal gas gets doubled. Consequently the mean collision time between the gas molecule changes from $\tau_{1}$ to $\tau_{2} .$ If $\frac{C_{p}}{C_{v}}=\gamma$ for this gas then a good estimate for $\frac{\tau_{2}}{\tau_{1}}$ is given by :View Solution
- 4A flask contains Hydrogen and Argon in the ratio $2: 1$ by mass. The temperature of the mixture is $30^{\circ} C$. The ratio of average kinetic energy per molecule of the two gases ( $K$ argon/ $K$ hydrogen) is: (Given: Atomic Weight of $Ar = 39.9$)View Solution
- 5View SolutionWhen air is filled in the balloon, the pressure and volume both increases while temperature does not change. Here Boyle's law is not obeyed because
- 6An object is placed in a medium of refractive index $3$. An electromagnetic wave of intensity $6 \times 10^8 \mathrm{~W} / \mathrm{m}^2$ falls normally on the object and it is absorbed completely. The radiation pressure on the object would be (speed of light in free space $=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ):View Solution
- 7An ideal gas occupies a volume of $2\, m^3$ at a pressure of $3\times10^6\, Pa$. The energy of the gas isView Solution
- 8View SolutionSelect the incorrect relation. (Where symbols have their usual meanings)
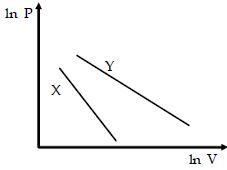
- 9For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution
- 10A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution