The specific heat of a gas in an isothermal process is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
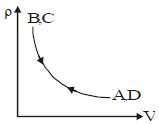
- 1Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
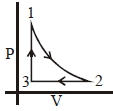
- 2An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
- 3One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
- 4View SolutionIn which thermodynamic process, volume remains same
- 5For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
- 6A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
- 7A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
- 8If $\gamma = 2.5$ and volume is equal to $\frac{1}{8}$ times to the initial volume then pressure $P' $ is equal to (Initial pressure $= P$)View Solution
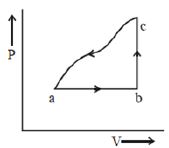
- 9A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 10Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution