A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$70$ calories of heat are required to raise the temperature of $2$ moles of an ideal gas at constant pressure from $30°C$ to $35°C.$ The amount of heat required to raise the temperature of same gas through the same range $(30°C$ to $35°C)$ at constant volume ..... $cal$ $(R = 2 \,cal/mol/K)$View Solution
- 2View SolutionThat gas cannot be liquified
- 3$\mathrm{N}$ moles of a polyatomic gas $(f=6)$ must be mixed with two moles of a monoatomic gas so that the mixture behaves as a diatomic gas. The value of $\mathrm{N}$ is:View Solution
- 4If the volume of the gas containing $n$ number of molecules is $V,$ then the pressure will decrease due to force of intermolecular attraction in the proportionView Solution
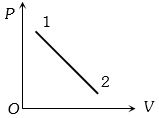
- 5A volume $V$ and pressure $P$ diagram was obtained from state $1$ to state $2$ when a given mass of a gas is subjected to temperature changes. During this process the gas isView Solution
- 6A cubical box with porous walls containing an equal number of ${O_2}$ and $H_2$ molecules is placed in a large evacuated chamber. The entire system is maintained at constant temperature $T.$ The ratio of ${v_{rms}}$ of ${O_2}$ molecules to that of the ${v_{rms}}$ of $H_2$ molecules, found in the chamber outside the box after a short interval isView Solution
- 7View SolutionAt the same temperature and pressure and volume of two gases, which of the following quantities is constant
- 8View SolutionAn ideal gas is filled in a closed container and container is moving with uniform acceleration in horizontal direction. Neglect gravity. Pressure inside the container is ...............
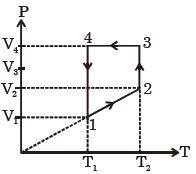
- 9A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 10A vessel contains $1$ mole of $O_2$ gas (molar mass $32$) at a temperature $T$. The pressure of the gas is $P$. An identical vessel containing one mole of $He$ gas (molar mass $4$) at a temperature $2T$ has a pressure ofView Solution
