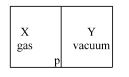
A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?

Medium
Work done by a gas is given by$:$
$w=\int P d V$
Work done by $x$ is against vacuum, hence $P=0$
$w=\int P d V=\int 0 d V=0$
Hence no work is done by or done on $x$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
- 2A gas expands $0.25{m^3}$ at constant pressure ${10^3}N/{m^2}$, the work done isView Solution
- 3A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution
- 4A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
- 5$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
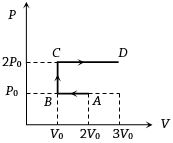
- 6$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ isView Solution
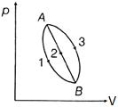
- 7An ideal gas of mass $m$ in a state $A$ goes to another state $B$ via three different processes as shown in figure. If $Q_{1}, Q_{2}$ and $Q_{3}$ denote the heat absorbed by the gas along the three paths, thenView Solution
- 8View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
- 9In an adiabatic process where in pressure is increased by $\frac{2}{3}\% $ if $\frac{{{C_p}}}{{{C_v}}} = \frac{3}{2},$ then the volume decreases by aboutView Solution
- 10Efficiency of Carnot engine is $100\%$ ifView Solution
