In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gas
IIT 2001, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
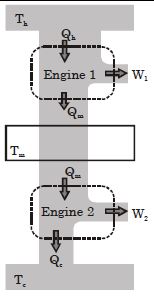
- 1Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\epsilon_1$ and $\epsilon_2$, respectively. The net efficiency of the combination is given by :View Solution
- 2The equation of state for a gas is given by $PV = nRT + \alpha V$, where $n$ is the number of moles and $\alpha $ is a positive constant. The initial temperature and pressure of one mole of the gas contained in a cylinder are $T_o$ and $P_o$ respectively. The work done by the gas when its temperature doubles isobarically will beView Solution
- 3Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 4The pressure and volume of an ideal gas are related as $\mathrm{PV}^{3 / 2}=\mathrm{K}$ (Constant). The work done when the gas is taken from state $A\left(P_1, V_1, T_1\right)$ to state $\mathrm{B}\left(\mathrm{P}_2, \mathrm{~V}_2, \mathrm{~T}_2\right)$ is :View Solution
- 5View SolutionFirst law thermodynamics states that
- 6$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
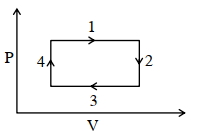
- 7An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution
- 8View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
- 9A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
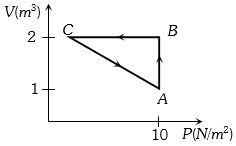
- 10An ideal gas is taken through the cycle $A → B → C → A$, as shown in the figure. If the net heat supplied to the gas in the cycle is $5 \ J$, the work done by the gas in the process $C → A$ is ....... $J$View Solution