==>${\eta _B} = \frac{{{T_2} - {T_3}}}{{{T_2}}} = \frac{{{W_B}}}{{{Q_2}}}$
$\therefore$ $\frac{{{Q_1}}}{{{Q_2}}} = \frac{{{T_1}}}{{{T_2}}} \times \frac{{{T_2} - {T_3}}}{{{T_1} - {T_2}}} = \frac{{{T_1}}}{{{T_2}}}$
$\therefore$ ${W_A} = {W_B}$
$\therefore$ ${T_2} = \frac{{{T_1} + {T_3}}}{2} = \frac{{800 + 300}}{2} = 550K$
Download our appand get started for free
Similar Questions
- 1A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process ..... $J$. ($J = 4.18$ joules/cal)View Solution
- 2The coefficient of performance of a refrigerator is $5$ . If the temperature inside freezer is $-20\,^oC$ , the temperature of the surroundings to which it rejects heat is ....... $^oC$View Solution
- 3A litre of dry air at $STP$ expands adiabatically to a volume of $3$ litres. If $\gamma=1.40,$ the work done by air is$(3^{1.4}=4.6555)$ [Take air to be an ideal gas $]$View Solution
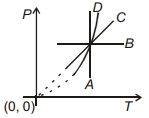
- 4Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution
- 5View SolutionA Camot cycle consists of
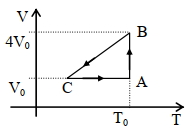
- 6One mole of an ideal gas in initial state $\mathrm{A}$ undergoes a cyclic process $A B C A$, as shown in the figure. Its pressure at $A$ is $\mathrm{P}_0$. Choose the correct option$(s)$ from the followingView Solution
$(A)$ Internal energies at $\mathrm{A}$ and $\mathrm{B}$ are the same
$(B)$ Work done by the gas in process $\mathrm{AB}$ is $\mathrm{P}_0 \mathrm{~V}_0 \ln 4$
$(C)$ Pressure at $C$ is $\frac{P_0}{4}$
$(D)$ Temperature at $\mathrm{C}$ is $\frac{\mathrm{T}_0}{4}$
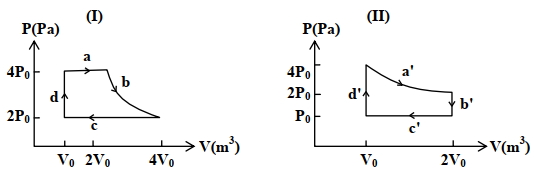
- 7One mole of an ideal gas undergoes two different cyclic processes I and II, as shown in the $P-V$ diagrams below. In cycle I, processes $a, b, c$ and $d$ are isobaric, isothermal, isobaric and isochoric, respectively. In cycle II, processes $a^{\prime}, b^{\prime}, c^{\prime}$ and $d^{\prime}$ are isothermal, isochoric, isobaric and isochoric, respectively. The total work done during cycle I is $W_I$ and that during cycle II is $W_{I I}$. The ratio $W_I / W_{I I}$ is . . . .View Solution
(image)
- 8View SolutionWhich of the following processes is reversible
- 9A mixture of ideal gas containing $5$ moles of monatomic gas and $1$ mole of rigid diatomic gas is initially at pressure $P _0$, volume $V _0$ and temperature $T _0$. If the gas mixture is adiabatically compressed to a volume $V _0 / 4$, then the correct statement(s) is/are,View Solution
(Give $2^{1.2}=2.3 ; 2^{3.2}=9.2 ; R$ is gas constant)
$(1)$ The final pressure of the gas mixture after compression is in between $9 P _0$ and $10 P _0$
$(2)$ The average kinetic energy of the gas mixture after compression is in between $18 RT _0$ and $19 RT _0$
$(3)$ The work $| W |$ done during the process is $13 RT _0$
$(4)$ Adiabatic constant of the gas mixture is $1.6$
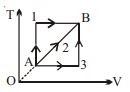
- 10A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution