The temperature below which a gas should be cooled, before it can be liquified by pressure only is termed as
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The plot that depicts the behavior of the mean free time $t$ (time between two successive collisions) for the molecules of an ideal gas, as a function of temperature $(T)$, qualitatively, is (Graphs are schematic and not drawn to scale)View Solution
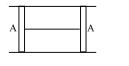
- 2A cylindrical tube of cross-sectional area $A$ has two air tight frictionless pistons at its two ends. The pistons are tied with a straight two ends. The pistons are tied with a straight piece of metallic wire. The tube contains a gas at atmospheric pressure $P_0$ and temperature $T_0$. If temperature of the gas is doubled then the tension in the wire isView Solution
- 3A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300\ K.$ The ratio of the average rotational kinetic energy per $O_2$ molecule to that per $N_2$ molecule isView Solution
- 4View SolutionFor an ideal gas of diatomic molecules
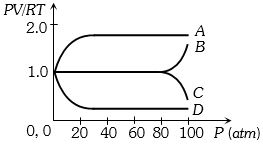
- 5An experiment is carried on a fixed amount of gas at different temperatures and at high pressure such that it deviates from the ideal gas behaviour. The variation of $\frac{{PV}}{{RT}}$ with $P$ is shown in the diagram. The correct variation will correspond toView Solution
- 6View SolutionSupposing the distance between the atoms of a diatomic gas to be constant, its specific heat at constant volume per mole (gram mole) is
- 7Let $\bar v , \bar v_{rms}$ and $v_p$ respectively denote the mean speed, root mean square speed and most probable speed of the molecules in an ideal monoatomic gas at absolute temperature $T$. The mass of the molecule is $m$. ThenView Solution
- 8A container holds $10^{26} molecules/m^3$ each of mass $3 \times 10^{-27}\,\,kg$. Assume that $1/6$ of the molecules move with velocity $2000 \,\,m/s$ directly towards one wall of the container while the remaining $5/6$ of the molecules move either away from the wall or in perpendicular direction, and all collisions of the molecules with the wall are elasticView Solution
- 9View SolutionVelocity of sound measured at a given temperature in oxygen and hydrogen is in the ratio
- 10Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent degree of freedom $f$ of the mixture isView Solution
