An experiment is carried on a fixed amount of gas at different temperatures and at high pressure such that it deviates from the ideal gas behaviour. The variation of $\frac{{PV}}{{RT}}$ with $P$ is shown in the diagram. The correct variation will correspond to
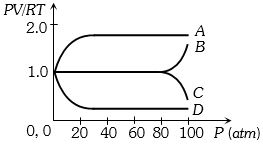

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A system consists of two types of gas molecules $A$ and $B$ having same number density $2 \times$ $10^{25}\, / {m}^{3}$. The diameter of ${A}$ and ${B}$ are $10\, \stackrel{\circ}{{A}}$ and $5\, \stackrel{\circ}{{A}}$ respectively. They suffer collision at room temperature. The ratio of average distance covererd by the molecule $A$ to that of $B$ between two successive collision is $.....\,\times 10^{-2}$View Solution
- 2In an ideal gas at temperature $T,$ the average force that a molecule applies on the walls of a closed container depends on $T$ as $T^q$ . A good estimate for $q$ isView Solution
- 3View SolutionMaxwell distribution function is shown in figure for different gases, which of the following is correct matching?
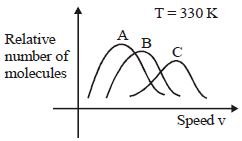
- 4The number density of molecules of a gas depends on their distance $r$ from the origin as, $n\left( r \right) = {n_0}{e^{ - \alpha {r^4}}}$. Then the total number of molecules is proportional toView Solution
- 5Aclosed vessel contains a mixture of two diatomic gases Aand $B$. Molar mass of Ais $16$ times that of $B$ and mass of gas $A$ contained in the vessel is $2$ times that of $B$. The following statements are givenView Solution
$(i)$Average kinetic energy per molecule of $A$ is equal to that of $B$.
$(ii)$ Root mean square value of translational velocity of $B$ is four times that of $A$.
$(iii)$ Pressure exerted by $B$ is eight times of that exerted by $A$.
$(iv)$ Number of molecules of $B$ in the cylinder is eight time that of $A$.
- 6A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$View Solution
- 7A mixture of $2$ moles of helium gas (atomic mass $=4 \ amu$ ), and $1$ mole of argon gas (atomic mass $=40 \ amu$ ) is kept at $300 \ K$ in a container. The ratio of the rms speeds $\left(\frac{\left. v _{ mms } \text { (helium }\right)}{ v _{\text {rms }} \text { (argon) }}\right)$ is :View Solution
- 8View SolutionAccording to kinetic theory of gases,
- 9Given below are two statements:View Solution
Statements $I:$ The temperature of a gas is $-73^{\circ}\,C$. When the gas is heated to $527^{\circ}\,C$, the root mean square speed of the molecules is doubled.
Statement $II:$ The product of pressure and volume of an ideal gas will be equal to translational kinetic energy of the molecules.
In the light of the above statements, choose the correct answer from the options given below :
- 10${O_2}$ gas is filled in a vessel. If pressure is doubled, temperature becomes four times. How many times its density will becomeView Solution
