The temperature inside a refrigerator is $t_2 \,^o C$ and the room temperature is $t_1\,^o C.$ The amount of heat delivered to the room for each joule of electrical energy consumed ideally will be
NEET 2016, Medium
Temperature inside refrigerator $ = {t_2}{\,^ \circ }C$
Room temperature $ = {t_1}{\,^ \circ }C$
For refrigerator,
$\frac{{Heat\,given\,to\,high\,temperature\,\left( {{Q_1}} \right)}}{{Heat\,taken\,from\,lower\,temperature\,\left( {{Q_2}} \right)}} = \frac{{{T_1}}}{{{T_2}}}$
$\frac{{{Q_1}}}{{{Q_2}}} = \frac{{{t_1} + 273}}{{{t_2} + 273}}$
$ \Rightarrow \frac{{{Q_1}}}{{{Q_1} - W}} = \frac{{{t_1} + 273}}{{{t_2} + 273}}\,\,or\,\,1 - \frac{W}{{{Q_1}}} = \frac{{{t_2} + 273}}{{{t_1} + 273}}$
$or\,\,\frac{W}{Q_1} = \frac{{{t_1} - {t_2}}}{{{t_1} + 273}}$
The amount of heat delivered to the room for each joule pf electrical energy $\left( {W = 1\,J} \right)$
${Q_1} = \frac{{{t_1} + 273}}{{{t_1} - {t_2}}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Starting with the same initial conditions, an ideal gas expands from volume $V_{1}$ to $V_{2}$ in three different ways. The work done by the gas is $W_{1}$ if the process is purely isothermal. $W _{2}$. if the process is purely adiabatic and $W _{3}$ if the process is purely isobaric. Then, choose the coned optionView Solution
- 2A diatomic ideal gas is used in a Carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\ V$, the efficiency of the engine isView Solution
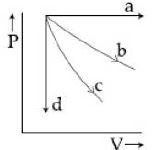
- 3View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 4View SolutionIf an ideal gas is compressed isothermally then
- 5A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
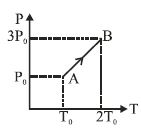
- 6Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
- 7One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
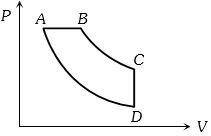
- 8An ideal gas is made to undergo a cycle depicted by the $p-V$ diagram given below. The curved line from $A$ to $B$ is an adiabat.Then,View Solution
- 9$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 10View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by