A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 2View SolutionA cycle tyre bursts suddenly. This represents an
- 3A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
- 4$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.View Solution
$Reason :$ The adiabatic compression is a slow process. - 5In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
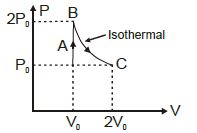
- 6The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution
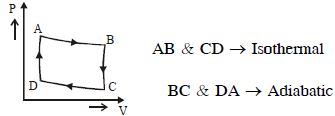
- 7View SolutionThe heat absorbed by a system in going through the given cyclic process is:
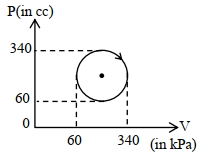
- 8Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
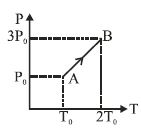
- 9The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 10View SolutionWhich of the following is a slow process
