The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.
AIIMS 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhen a gas expands adiabatically
- 2A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process ..... $J$. ($J = 4.18$ joules/cal)View Solution
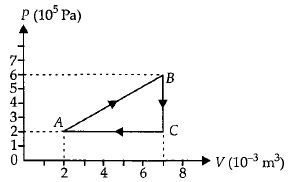
- 3A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 4The efficiency of a Carnot engine operating with reservoir temperature of $100\,^{\circ} C$ and $-23\,^{\circ} C$ will beView Solution
- 5The temperature inside and outside a refrigerator are $273 \,K$ and $300 \,K$ respectively. Assuming that the refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be nearly ........ $J$View Solution
- 6The work of $146\,kJ$ is performed in order to compress one kilo mole of a gas adiabatically and in this process the temperature of the gas increases by $7\,^oC$ . The gas is $(R = 8.3\, J\, mol^{-1}\, K^{-1})$View Solution
- 7Air is pumped into a balloon, of initial volume $V$ , until its diameter has doubled. If the atmospheric pressure is $p$ , what is the work done against the atmosphere ?View Solution
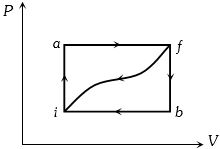
- 8When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$View Solution
- 9$1\,kg$ of water at $100\, ^{\circ}C$ is converted into steam at $100^{\circ}\,C$ by boiling at atmospheric pressure. The volume of water changes from $1.00 \times 10^{-3}\,m ^3$ as a liquid to $1.671\,m ^3$ as steam. The change in internal energy of the system during the process will be $........kJ$ (Given latent heat of vaporisaiton $=2257\,kJ / kg$. Atmospheric pressure $=1 \times 10^5\,Pa$ )View Solution
- 10View SolutionIn thermodynamics, heat and work are
