The work done in an adiabatic change in a gas depends only on
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
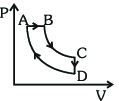
- 1A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution
- 2The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
- 3Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 4Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
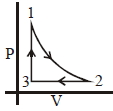
- 5Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 6View SolutionBy opening the door of a refrigerator placed inside a room you
- 7Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
- 8View SolutionA perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
- 9The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.View Solution
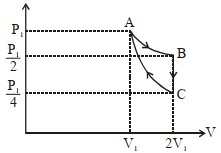
- 10If one mole of an ideal gas at $\left( P _{1}, V _{1}\right)$ is allowed to expand reversibly and isothermally ($A$ to $B$ ) its pressure is reduced to one-half of the original pressure (see $figure$). This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value $( B \rightarrow C ) .$ Then it is restored to its initial state by a reversible adiabatic compression ($C$ to $A$). The net workdone by the gas is equal to ...... .View Solution