The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.
AIIMS 2019, Medium
The efficiency is given as,
$\eta=1-\frac{ T _{2}}{ T _{1}}$
Substitute the values.
$\eta=1-\frac{273+4}{273+15}$
$=0.038$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ The Carnot cycle is useful in understanding the performance of heat engines.View Solution
$Reason :$ The Carnot cycle provides a way of determining the maximum possible efficiency achievable with reservoirs of given temperatures. - 2View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
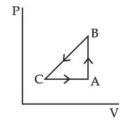
- 3The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
- 4Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 5A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$View Solution
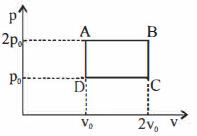
- 6The above $P-V$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle isView Solution
- 7View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
- 8If during an adiabatic process the pressure of mixture of gases is found to be proportional to square of its absolute temperature. The ratio of $C_p / C_v$ for mixture of gases is .........View Solution
- 9A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
- 10A sample of an ideal gas is taken through the cyclic process $ABCA$ as shown in figure. It absorbs, $40\,J$ of heat during the part $A B$, no heat during $BC$ and rejects $60\,J$ of heat during $CA$. $A$ work $50\,J$ is done on the gas during the part $BC$. The internal energy of the gas at $A$ is $1560\,J$. The work done by the gas during the part $CA$ is.............$J$View Solution