Even Carnot engine cannot give $100\%$ efficiency because we cannot
AIEEE 2002, Medium
(c) The efficiency of Carnot engine is $\quad \eta=1-\frac{T_{2}}{T_{1}}$
where, $T_{1}$ is the temperature of the source and $T_{2}$ that
of sink. since, $\frac{T_{2}}{T_{1}}=\frac{Q_{2}}{Q_{1}}$
So, $\quad \eta=1-\frac{Q_{2}}{Q_{1}} |$
To obtain $100 \%$ efficiency (i.e., $\eta=1), Q_{2}$ must be zero i.e., if a sink at absolute zero would be available, all the heat taken from the source would have been converted into work. The temperature of sink means a negative temperature on the absolute scale at which the efficiency of engine is greater than unity. This would be a violation of the $2\, nd$ law of thermodynamics. Hence, a negative temperature on the absolute scale is impossible. Hence, we cannot reach absolute zero temperature.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
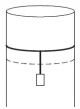
- 2A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 3A litre of dry air at $STP$ expands adiabatically to a volume of $3$ litres. If $\gamma=1.40,$ the work done by air is$(3^{1.4}=4.6555)$ [Take air to be an ideal gas $]$View Solution
- 4View SolutionIn adiabatic expansion of a gas
- 5View SolutionIf an ideal gas is compressed isothermally then
- 6$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
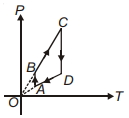
- 7A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
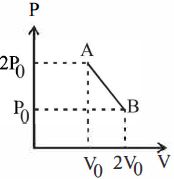
- 8'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will beView Solution
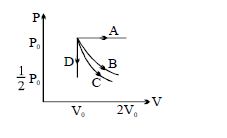
- 9The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution
- 10A cylinder with a movable piston contains $3\,moles$ of hydrogen at standard temperature and pressure. The walls of the cylinder are made of a heat insulator, and the piston is insulated by having a pile of sand on it. By what factor does the pressure of the gas increases if the gas is compressed to half its original volume?View Solution
