By opening the door of a refrigerator placed inside a room you
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
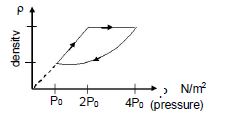
- 2An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 3The temperature inside and outside of refrigerator are $260\, K$ and $315\, K$ respectively. Assuming that the refrigerator cycle is reversible, calculate the heat delivered to surroundings for every joule of work done.View Solution
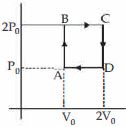
- 4Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
- 5A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
- 6This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.Statement $2 :$ The temperature of a gas remains constant in an adiabatic process.
- 7View SolutionIn isothermic process, which statement is wrong
- 8View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
- 9A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 10During which of the following thermodynamic process represented by $P V$ diagram the heat energy absorbed by system may be equal to area under $P V$ graph?View Solution
