Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
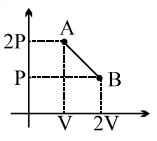
- 2One mole of a gas expands obeying the relation as shown in the $P/V$ diagram. The maximum temperature in this process is equal toView Solution
- 3A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
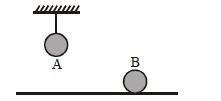
- 4Two identical balls, $A$ and $B$ , of uniform composition and initially at the same temperature, each absorb exactly the same amount of heat. $A$ is hanging down from the ceiling while $B$ rests on the horizontal floor in the same room. Assuming no subsequent heat loss by the balls, which of the following statements is correct about their final temperatures, $T_A$ and $T_B$ , once the balls have reached their final state?View Solution
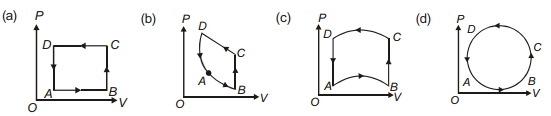
- 5In following figures $(a)$ to $(d)$, variation of volume by change of pressure is shown in figure. The gas is taken along the path $A B C D A$. Change in internal energy of the gas will be .......View Solution
- 6Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 7$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 8The amount of heat needed to raise the temperature of $4\, moles$ of a rigid diatomic gas from $0^{\circ} {C}$ to $50^{\circ} {C}$ when no work is done is ......${R}$ ($R$ is the universal gas constant)View Solution
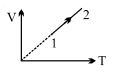
- 9An ideal gas undergoes the process $1 \rightarrow 2$ as shown in the figure, the heat supplied and work done in the process is $\Delta \,\,Q$ and $\Delta \,\,W$ respectively. The ratio $\Delta \,\,Q :$ $\Delta \,\,W$ isView Solution
- 10View SolutionOut of the following which quantity does not depend on path
