Two cylinders contain same amount of ideal monatomic gas. Same amount of heat is given to two cylinders. If temperature rise in cylinder $A$ is $T_0$ then temperature rise in cylinder $B$ will be .........
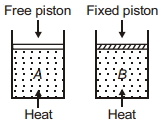

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
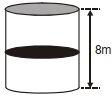
- 2A thermally isolated cylindrical closed vessel of height $8 m$ is kept vertically. It is divided into two equal parts by a diathermic (perfect thermal conductor) frictionless partition of mass $8.3 kg$. Thus the partition is held initially at a distance of $4 m$ from the top, as shown in the schematic figure below. Each of the two parts of the vessel contains $0.1$ mole of an ideal gas at temperature $300 K$. The partition is now released and moves without any gas leaking from one part of the vessel to the other. When equilibrium is reached, the distance of the partition from the top (in $m$ ) will be. . . . . . (take the acceleration due to gravity $=10 ms ^{-2}$ and the universal gas constant $=8.3 J mol ^{-1} K ^{-1}$ ).View Solution
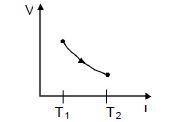
- 3The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)View Solution
- 4Given that $1\,g$ of water in liquid phase has volume $1\,cm^3$ and in vapour phase $1671\, cm^3$ at atmospheric pressure and the latent heat of vaporization of water is $2256\,J/g;$ the change in the internal energy in joules for $1\,g$ of water at $373\,K$ when it changes from liquid phase to vapour phase at the same temperature is ....... $J$View Solution
- 5An ideal gas at pressure $P$ and volume $V$ is expanded to volume$ 2V.$ Column $I$ represents the thermodynamic processes used during expansion. Column $II$ represents the work during these processes in the random order.:View Solution
Column $I$ Column $II$ $(p)$ isobaric $(x)$ $\frac{{PV(1 - {2^{1 - \gamma }})}}{{\gamma - 1}}$ $(q)$ isothermal $(y)$ $PV$ $(r)$ adiabatic (z) $PV\,\iota n\,2$ The correct matching of column $I$ and column $II$ is given by
- 6The coefficient of performance of a refrigerator is $5$ . If the temperature inside freezer is $-20\,^oC$ , the temperature of the surroundings to which it rejects heat is ....... $^oC$View Solution
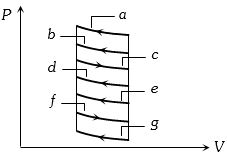
- 7The $P-V$ diagram shows seven curved paths (connected by vertical paths) that can be followed by a gas. Which two of them should be parts of a closed cycle if the net work done by the gas is to be at its maximum valueView Solution
- 8$Assertion :$ The Carnot cycle is useful in understanding the performance of heat engines.View Solution
$Reason :$ The Carnot cycle provides a way of determining the maximum possible efficiency achievable with reservoirs of given temperatures. - 9View SolutionIn isothermal expansion, the pressure is determined by
- 10A van der Waal's gas obeys the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$. Its internal energy is given by $U=C T-\frac{n^2 a}{V}$. The equation of a quasistatic adiabat for this gas is given byView Solution
