$\mathrm{COP}=\frac{\mathrm{T}_{\mathrm{L}}}{\mathrm{T}_{\mathrm{H}}-\mathrm{T}_{\mathrm{L}}}$
Where $\mathrm{T}_{\mathrm{L}} \rightarrow$ lower Temperature
$\mathrm{\&} \quad \mathrm{T}_{\mathrm{H}} \rightarrow$ Higher Temperature
So, $5=\frac{T_{L}}{T_{H}-T_{L}}$
$\Rightarrow \mathrm{T}_{\mathrm{H}}=\frac{6}{5} \mathrm{T}_{\mathrm{L}}=\frac{6}{5}(253)=303.6 \mathrm{K}$
Download our appand get started for free
Similar Questions
- 1If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 2$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 3The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
- 4One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
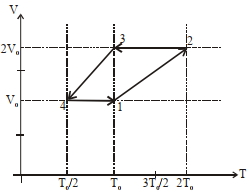
- 5One mole of a monoatomic ideal gas goes through a thermodynamic cycle, as shown in the volume versus temperature ($V-T$) diagram. The correct statement($s$) is/are :View Solution
[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$
- 6A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
- 7The temperature inside and outside of refrigerator are $260\, K$ and $315\, K$ respectively. Assuming that the refrigerator cycle is reversible, calculate the heat delivered to surroundings for every joule of work done.View Solution
- 8View SolutionThe internal energy of the gas increases In
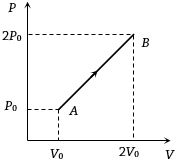
- 9The $P-V$ diagram of $2$ gm of helium gas for a certain process $A \to B$ is shown in the figure. what is the heat given to the gas during the process $A \to B$View Solution
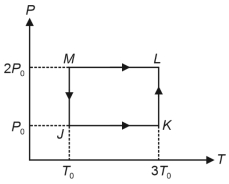
- 10One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P - T$ diagram.View Solution
Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
$List-I$ $List-II$ ($P$) Work done in the complete cyclic process ($1$) $R T_0-4 \ R T_0 \ln 2$ ($Q$) Change in the internal energy of the gas in the process $JK$ ($2$) $0$ ($R$) Heat given to the gas in the process $KL$ ($3$) $3 \ R T_0$ ($S$) Change in the internal energy of the gas in the process $MJ$ ($4$) $-2 \ R T_0 \ln 2$ ($5$) $-3 \ R T_0 \ln 2$