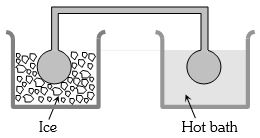
Two identical glass bulbs are interconnected by a thin glass tube. A gas is filled in these bulbs at $N.T.P.$ If one bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes $1.5$ times. The temperature of hot bath will be ....... $^oC$

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When a gas in a closed vessel was heated so as to increase its temperature by ${5^o}C$, there occurred an increase of $1\%$ in its pressure. The original temperature of the gas was ...... $^oC$View Solution
- 2A sample of gas is at $0°C.$ To what temperature it must be raised in order to double the $r.m.s.$ speed of the molecule ....... $^oC$View Solution
- 3The average thermal energy for a mono$-$atomic gas is : $\left( k _{ B }\right.$ is Boltzmann constant and $T ,$ absolute $e$. temperature)View Solution
- 4At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
- 5According to the kinetic theory of gases the $r.m.s.$ velocity of gas molecules is directly proportional toView Solution
- 6If the degree of freedom of a gas are $f,$ then the ratio of two specific heats ${C_P}/{C_V}$ is given byView Solution
- 7View SolutionFor matter to exist simultaneously in gas and liquid phases
- 8$0.056 \,kg$ of Nitrogen is enclosed in a vessel at a temperature of $127\,^{\circ} C$. The amount of heat required to double the speed of its molecules is k cal. (Take $R =2$ $cal \,mole$ $^{-1} K ^{-1}$ )View Solution
- 9In the relation $n = \frac{{PV}}{{RT}},n = $View Solution
- 10At a temperature $T K,$ the pressure of $4.0\,\,g$ argon in a bulb is $p$. The bulb is put in a bath having temperature higher by $50\,\,K$ than the first one. $0.8\,\,g$ of argon gas had to be removed to maintained original pressure. The temperature $T$ is equal to ..... $K$View Solution
