When a gas in a closed vessel was heated so as to increase its temperature by ${5^o}C$, there occurred an increase of $1\%$ in its pressure. The original temperature of the gas was ...... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An insulated box containing a diatomic gas of molar mass $M$ is moving with a velocity $v$. The box is suddenly stopped. The resulting change in temperature isView Solution
- 2The temperature of an ideal gas is reduced from $927^\circ C$ to $27^\circ C$. The $r.m.s.$ velocity of the molecules becomesView Solution
- 3View SolutionThe root mean square speed of the molecules of a gas is
- 4$Assertion :$ One mole of any substance at any temperature or volume always contains $6.02\times10^{23}$ molecules.View Solution
$Reason :$ One mole of a substance always refers to $S.T.P.$ conditions. - 5A box contains a mixture of ${H_2}$ and $He$ gases. Which of the following statements are correctsView Solution
- 6The temperature of a gas having $2.0 \times 10^{25}$ molecules per cubic meter at $1.38 \mathrm{~atm}$ (Given, $\mathrm{k}=$ $\left.1.38 \times 10^{-23} \mathrm{JK}^{-1}\right)$ is :View Solution
- 7If pressure of $C{O_2}$ (real gas) in a container is given by $P = \frac{{RT}}{{2V - b}} - \frac{a}{{4{b^2}}}$ then mass of the gas in container is ...... $gm$View Solution
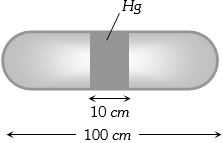
- 8A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 9View SolutionIn which of the following gas the root mean square velocity will be minimum (at const. temperature)
- 10One mole of an ideal monatomic gas undergoes a process described by the equation $PV^3 =$ constant. The heat capacity of the gas during this process isView Solution
