The average thermal energy for a mono$-$atomic gas is : $\left( k _{ B }\right.$ is Boltzmann constant and $T ,$ absolute $e$. temperature)
NEET 2020, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider a gas with density $\rho $ and $\bar c$ as the root mean square velocity of its molecules contained in a volume. If the system moves as whole with velocity $v,$ then the pressure exerted by the gas isView Solution
- 2View SolutionAccording to law of equipartition of energy the molar specific heat of a diatomic gas at constant volume where the molecule has one additional vibrational mode is :-
- 3View SolutionSelect the incorrect relation. (Where symbols have their usual meanings)
- 4A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 5If $r.m.s.$ velocity of a gas is ${V_{rms}} = 1840m/s$ and its density $\rho = 8.99 \times {10^{ - 2}}kg/{m^3},$ the pressure of the gas will beView Solution
- 6$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
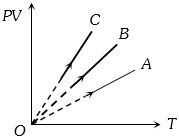
- 7$2\, gm$ of ${O_2}$ gas is taken at $27°C$ and pressure $76\, cm.$ Of $Hg$. Then find out volume of gas (in litre)View Solution
- 8A hydrogen cylinder is designed to withstand an internal pressure of $100 \,atm$. At $27^{\circ} C$, hydrogen is pumped into the cylinder which exerts a pressure of $20 \,atm$. At what temperature does the danger of explosion first sets in ......... $K$View Solution
- 9The given diagram shows isotherms for a fixed mass of an ideal gas at temperature $T_1$ and $T_2$ . What is the value of the ratio $\frac{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_{2{\text{ }}}}}}{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_1}}}$ ?View Solution
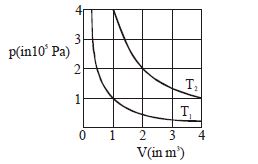
- 10The temperature of a gas is $ -68^\circ C$. At ...... $^oC$ temperature will the average kinetic energy of its molecules be twice that of at $ -68^\circ C$View Solution
