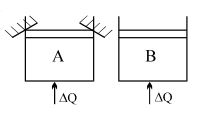
Two identical vessels $A \& B$ contain equal amount of ideal monoatomic gas. The piston of $A$ is fixed but that of $B$ is free. Same amount of heat is absorbed by$A \& B$. If $B'$s internal energy increases by $100 \,\,J$ the change in internal energy of $A$ is ...... .$J$

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monoatomic gas at pressure $P$ and volume $V$ is suddenly compressed to one eighth of its original volume. The final pressure at constant entropy will be $.....P$View Solution
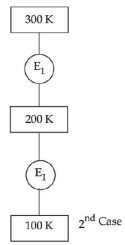
- 2In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
- 3One gm mol of a diatomic gas $(\gamma = 1.4)$ is compressed adiabatically so that its temperature rises from ${27^o}C$ to ${127^o}C$. The work done will beView Solution
- 4Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
- 5Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
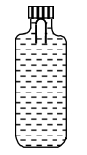
- 6A soft plastic bottle, filled with water of density $1 gm / cc$, carries an inverted glass test-tube with some air (ideal gas) trapped as shown in the figure. The test-tube has a mass of $5 gm$, and it is made of a thick glass of density $2.5 gm / cc$. Initially the bottle is sealed at atmospheric pressure $p_0=10^5 Pa$ so that the volume of the trapped air is $v_0=3.3 cc$. When the bottle is squeezed from outside at constant temperature, the pressure inside rises and the volume of the trapped air reduces. It is found that the test tube begins to sink at pressure $P_0+\Delta p$ without changing its orientation. At this pressure, the volume of the trapped air is $v_0-\Delta v$.View Solution
Let $\Delta v=X$ cc and $\Delta p=Y \times 10^3 Pa$.
($1$) The value of $X$ is
($2$) The value of $Y$ is
Give the answer or quetion ($1$) and ($2$)
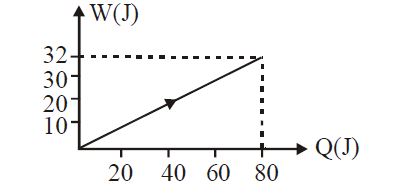
- 7View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
- 8Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
- 9View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 10For adiabatic processes $\left( {\gamma = \frac{{{C_p}}}{{{C_v}}}} \right)$View Solution
