A monoatomic gas at pressure $P$ and volume $V$ is suddenly compressed to one eighth of its original volume. The final pressure at constant entropy will be $.....P$
JEE MAIN 2022, Diffcult
Constant entropy means process is adiabatic
$PV ^{\gamma}=$ constant
$V _{2}=\frac{ V _{1}}{8}$
$P _{1} V _{1}^{\gamma}= P _{2} V _{2}^{\gamma}$
$P _{1} V _{1}^{\gamma}= P _{2}\left(\frac{ V _{1}}{8}\right)^{5 / 3}$
$P _{1} V _{1}^{5 / 3}=\frac{ P _{2} V _{1}^{5 / 3}}{32}$
$P _{2}=32 P _{1}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
- 2View SolutionDuring the thermodynamic process shown in figure for an ideal gas
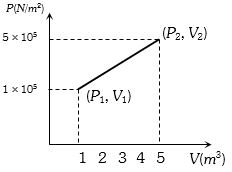
- 3A system changes from the state $({P_1},\;{V_1})$ to $({P_2},{V_2})$ as shown in the figure. What is the work done by the systemView Solution
- 4View SolutionOne mole of a monoatomic gas and one mole of a diatomic gas are initially in the same state. Both gases are expanded isothermally and then adiabatically, such that they acquire the same final state. Choose the correct statement.
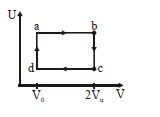
- 5The figure given below shows the variation in the internal energy $U$ with volume $V$ of $2.0\ mole$ of an ideal gas in a cyclic process $abcda$ . The temperatures of the gas during the processes $ab$ and $cd$ are $500\ K$ and $300\ K$ respectively, the heat absorbed by the gas during the complete process is .... $J$View Solution
(Take $R$ = $8.3\ J/mol-K$ and $ln\ 2$ = $0.69$)
- 6$0.02\, moles$ of an ideal diatomic gas with initial temperature $20^{\circ} C$ is compressed from $1500 \,cm ^{3}$ to $500 \,cm ^{3}$. The thermodynamic process is such that $p V^{2}=\beta$, where $\beta$ is a constant. Then, the value of $\beta$ is close to (the gas constant, $R=8.31 \,J / K / mol$ ).View Solution
- 7View SolutionThe isothermal bulk modulus of a perfect gas at normal pressure is
- 8One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
- 9View SolutionWork done on or by a gas, in general depends upon the
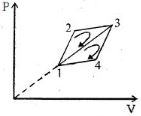
- 10The efficiency of a thermodynamic cycle $1-2-3- 1 ($see picture$)$ is $20\%$ and for another thermodynamic cycle $1 - 3-4 - 1$ efficiency is equal to $10\%$. Determine the efficiency $\eta ($ in $\%)$ of the thermodynamic cycle $1-2-3-4- 1.$The gas is assumed to be idealView Solution