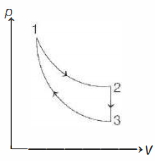
$\Delta W_{1}=\Delta W_{2}$ $\Rightarrow \Delta Q_{1}-\Delta U_{1}=\Delta Q_{2}-\Delta U_{2}$
$\Rightarrow \Delta Q_{1}-\Delta Q_{2}=\Delta U_{1}-\Delta U_{2}$
So, option $a$ is correct.
Process is isobaric
Hence $\Delta Q_{1}=n C_{p 1} \Delta T_{1}=\frac{5}{2} n R \Delta T_{1}=\frac{5}{2} P_{0} V_{0}$
Also $\Delta Q_{2}=n C_{P 2} \Delta T_{2}=\frac{7}{2} n R \Delta T_{2}=\frac{7}{2} P_{0} V_{0}$
Hence $\Delta Q_{2}>\Delta Q_{1}$
$\Rightarrow \Delta U_{1}+\Delta W_{1}>\Delta U_{2}+\Delta W_{2}$
So, option $b$ is correct
$\Delta U_{1}=n C_{v 1} \Delta T_{1}=\frac{3}{2} n R \Delta T_{1}=\frac{3}{2} P_{0} V_{0}$
$\Delta U_{2}=n C_{v 2} \Delta T_{2}=\frac{5}{2} n R \Delta T_{2}=\frac{5}{2} P_{0} V_{0}$
Hence $\Delta U_{2}>\Delta U_{1}$
So, option $c$ is also correct
Download our appand get started for free
Similar Questions
- 1Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
- 2One mole of an ideal gas at an initial temperature of $T\, K$ does $6 R$ joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$, the final temperature of gas will beView Solution
- 3A total of $48 \mathrm{~J}$ heat is given to one mole of helium kept in a cylinder. The temperature of helium increases by $2^{\circ} \mathrm{C}$. The work done by the gas is : (Given, $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$.)View Solution
- 4In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 5View SolutionIn an isothermal expansion
- 6$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.View Solution
$Reason :$ The adiabatic compression is a slow process. - 7In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 8Efficiency of Carnot engine is $100\%$ ifView Solution
- 9One mole of an ideal gas at an initial temperature of $T\, K$ does $6\, R\, joules$ of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $\frac{5}{3}$ , the final temperature of gas will beView Solution
- 10The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution