What is the correct order of reactivity of alcohols in the following reaction?
$\text{R}-\text{OH}+\text{HCL}\xrightarrow{\text{ZnCl}_2}\text{R}-\text{Cl}+\text{H}_2\text{O}$
$\text{R}-\text{OH}+\text{HCL}\xrightarrow{\text{ZnCl}_2}\text{R}-\text{Cl}+\text{H}_2\text{O}$
- 3° > 2° > 1°
This reaction is a type of nucleophilic substitution reaction in which -OH group is replaced by -Cl. The intermediate formed by tertiary alcohol is more stable as carbocation is more stable than primary and secondary carbocation. Thus, the order of reactivity of alcohol will be 3° > 2° > 1°.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe process of converting alkyl halides into alcohols involves_____________.
- 2View SolutionWhat is the functional group in alcohol?
- 3View SolutionArrange the following alkanols 1, 2 and 3 in order of their reactivity towards acid catalyzed dehydration.
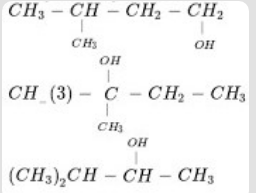
- 4The high boiling point of ethanol $(78.2^\circ C)$ compared to dimethyl ether $(−23.6^\circ C),$ though both having the same molecular formula $C_2H_6O,$ is due to:View Solution
- 5View SolutionIUPAC name of phenol is -
- 6When $3-$methylbutan$-2-$ol is treated with $\ce{HBr},$ the following reaction takes place. What are the correct steps for formation of the above product?View Solution
- 7$\text{CH}_3\text{−CH}\text{−CH}−\text{CH}_3\\ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \text{OH}\ \ \ \ \ \ \text{OH}$IUPAC name of the given compound is:View Solution
- 8The only alcohol that can be prepared by the indirect hydration of alkene is$:$View Solution
- 9View SolutionMaximum boiling point would be shown by:
- 10View SolutionWhich of the following reactions will yield phenol?
