Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
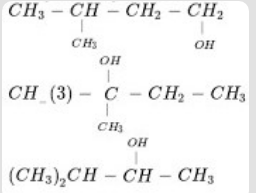
- 1View SolutionArrange the following alkanols 1, 2 and 3 in order of their reactivity towards acid catalyzed dehydration.
- 2View SolutionButane has a _______ boiling point to that of propanol.
- 3Ether is formed when alkyl halide is treated with sodium alkoxide. This method is known as$:$View Solution
- 4The high boiling point of ethanol $(78.2^\circ C)$ compared to dimethyl ether $(−23.6^\circ C),$ though both having the same molecular formula $C_2H_6O,$ is due to:View Solution
- 5View SolutionTrivial name of methanol is _________.
- 6View SolutionThe strongest acid in the following compounds is:
- 7The general formula for alcohol series is $\ce{C_nH_{2n+1}OH}.$ Which is the formula for alcohol contains four carbon?View Solution
- 8View SolutionThe correct order of boiling point for primary (1), secondary (2) and tertiary (3) alcohols is:
- 9During dehydration of alcohols to alkenes by heating with conc. $\ce{H_2SO_{4}},$ the initiation step is$:$View Solution
- 10View SolutionEthanol on reaction with acetic anhydride gives:
