What is the nature of change in internal energy in the following three thermodynamical processes shown in figure
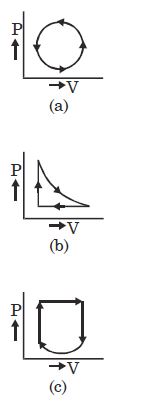

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
- 2An ideal gas at a pressures of $1$ atmosphere and temperature of ${27^o}C$ is compressed adiabatically until its pressure becomes $8$ times the initial pressure, then the final temperature is ..... $^oC$ ($\gamma = 3/2$)View Solution
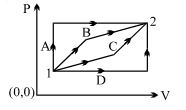
- 3An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
- 4A diatomic gas $(\gamma=1.4)$ does $200 \mathrm{~J}$ of work when it is expanded isobarically. The heat given to the gas in the process is:View Solution
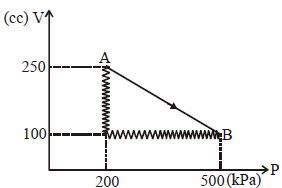
- 5A monoatomic gas is taken along path $AB$ as shown. Calculate change in internal energy of systemView Solution
- 6$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 7View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 8In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 9Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is ${m_A}$ and that in $B$ is ${m_B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2V$. The changes in the pressure in $A$ and $B$ are found to be $\Delta P$ and $1.5 \Delta P$ respectively. ThenView Solution
- 10View SolutionWhen heat in given to a gas in an isobaric process, then
