When $1\, gm$ of water at ${0^o}C$ and $1 \times {10^5}\;N/{m^2}$ pressure is converted into ice of volume $1.091\;c{m^2}$, the external work done will be
Easy
(a) It is an isothermal process. Hence work done $ = P({V_2} - {V_1})$
$ = 1 \times {10^5} \times (1.091 - 1) \times {10^{ - 6}} = 0.0091\;J$
$ = 1 \times {10^5} \times (1.091 - 1) \times {10^{ - 6}} = 0.0091\;J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
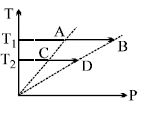
- 1On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
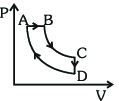
- 2A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution
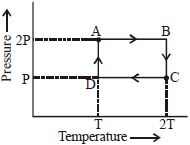
- 3An ideal monoatomic gas is taken through the thermodynamic states $A \to B \to C \to D$ via the paths shown in the figure. If $U_A, U_B, U_C$ and $U_D$ represent the internal energy of the gas in state $A, B\, C$ and $D$ respectively, then which of the following is not true?View Solution
- 4The amount of heat needed to raise the temperature of $4\, moles$ of a rigid diatomic gas from $0^{\circ} {C}$ to $50^{\circ} {C}$ when no work is done is ......${R}$ ($R$ is the universal gas constant)View Solution
- 5View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 6A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution
- 7In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 8View SolutionIn thermodynamic processes which of the following statements is not true?
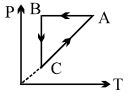
- 9A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
- 10An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
