An ideal monoatomic gas is taken through the thermodynamic states $A \to B \to C \to D$ via the paths shown in the figure. If $U_A, U_B, U_C$ and $U_D$ represent the internal energy of the gas in state $A, B\, C$ and $D$ respectively, then which of the following is not true?
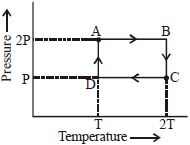

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn the adiabatic compression, the decrease in volume is associated with
- 2A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
- 3$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion. - 4A monoatomic gas is taken along path $AB$ as shown. Calculate change in internal energy of systemView Solution
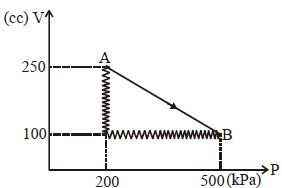
- 5View SolutionThe maximum possible efficiency of a heat engine is ...........
- 6If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, isView Solution
- 7The process $C D$ is shown in the diagram. As system is taken from $C$ to $D$, what happens to the temperature of the system?View Solution
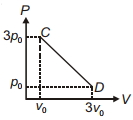
- 8View SolutionFor adiabatic process, wrong statement is
- 9View SolutionPressure versus temperature graph of an ideal gas is shown in figure
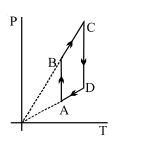
- 10A solid body of constant heat capacity $1\,J /{ }^{\circ} C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with 8 reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^{\circ}\,C$ to final temperature $200^{\circ}\,C$. Entropy change of the body in the two cases respectively is :
